Covid-19 In-Home Test Kits
Wholesale IgM/IgG Anti-Body Covid-19 Test Kits
SARS-CoV-2 (Covid-19) Antibody IgM/IgG Test Kits
InnoVita collaborates with innovators from around the world to develop new technologies and products for the detection and elimination of the SARS-CoV-2 virus and other airborne pathogens.

Product: Novel Coronavirus (2019-nCoV) IgM/IgG AntibodyCombo Test Kit (Colloidal Gold)
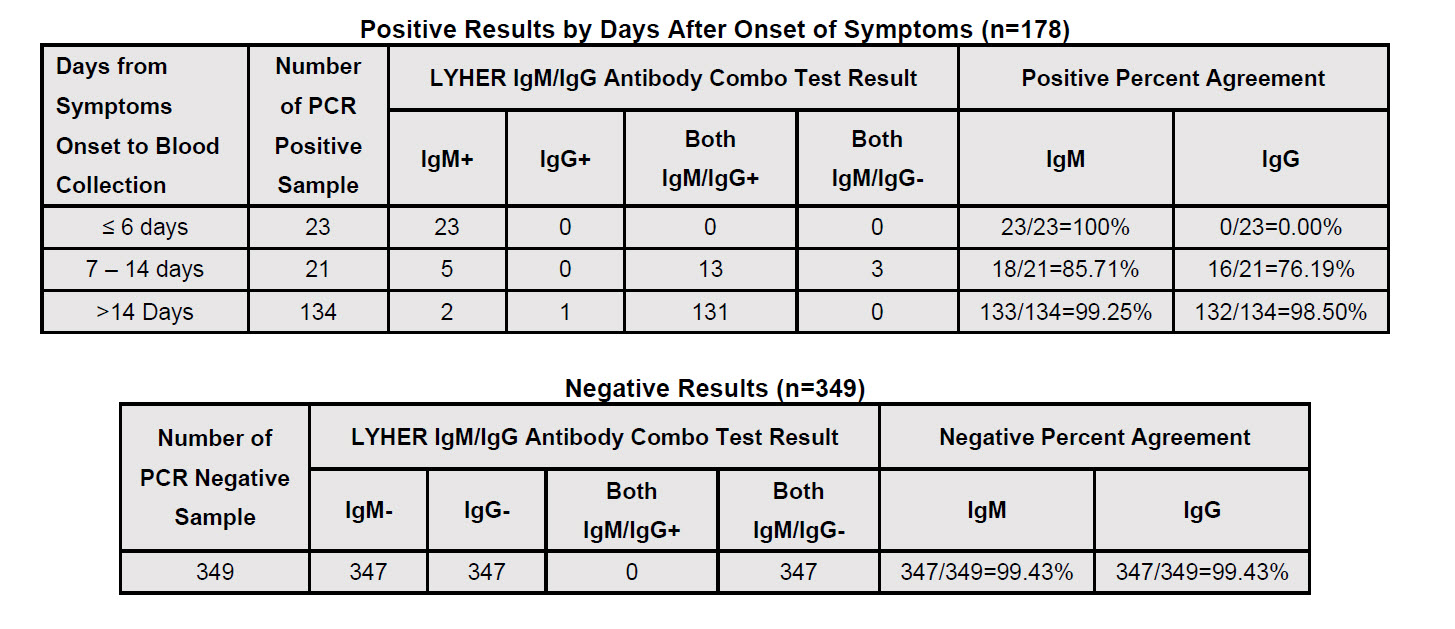
FDA Emergency Use Authorization (EUA): YES
The LYHER Novel Coronavirus (2019-nCoV) IgM/IgG Antibody Combo Test Kit (Colloidal Gold) is a lateral flow immunoassay intended for the qualitative detection of and differentiation of IgG and IgM antibodies to SARS-CoV-2 in serum and plasma (dipotassium-EDTA, lithium-heparin, or sodium-citrate).
The LYHER Novel Coronavirus (2019-nCoV) IgM/IgG Antibody Combo Test Kit (Colloidal Gold) is intended for use as an aid in identifying individuals with an adaptive immune response to SARS-CoV-2, indicating recent or prior infection.
The LYHER® Kit uses an immunochromatography based technique in which a colloidal gold-labeled antibody is used as a tracer to detect antibodies. The test panel contains (1) one colloidal gold-labeled recombinant COVID-19 S1 spike protein antigen and quality control antibody colloidal gold marker, (2) two detection lines: G and M, (3) one quality control line: C.
The test area of nitrocellulose membrane is coated with mouse anti-human-IgM monoclonal antibody, mouse anti-human-IgG monoclonal antibody and goat anti-rabbit antibody were immobilized at test M, G, and C, respectively. When an appropriate amount of test sample is loaded to LYHER® Kit, it will move along the nitrocellulose membrane.
If the specimen contains specific IgM/IgG antibodies against COVID-19, they will be captured by the mouse anti-human IgM/IgG monoclonal antibodies coated in the test area to form a visible strip. If neither antibody is present, a negative result is displayed. The Control Line is used as a procedural control. The control line should always appear if the test procedure is performed properly and the reagents are working as intended.
Our partner typically has 500k+ test kits in inventory in the US, with the ability to send up to 5 million more Covid-19 test kits within one week. They also expect to have Emergency Use Authorization (EUA) from the FDA for an Antigen in-home test kit within a few weeks.
Please complete the contact form below for additional information and pricing.






